If you're running a cannabis or hemp extraction operation, understanding decarboxylation isn't optional – it's essential. This chemical process transforms inactive acidic cannabinoids (THCA, CBDA) into their active forms (THC, CBD), directly impacting your product's potency, quality, and market value.
Table of Contents
ToggleIn this comprehensive guide, we'll break down the science of decarboxylation, share practical temperature and timing protocols, and reveal how your extraction method choice affects the entire process.
What is Decarboxylation?
Decarboxylation is a chemical reaction that removes a carboxyl group (-COOH) from cannabinoid molecules, releasing carbon dioxide (CO₂) in the process. In cannabis processing, this occurs when plant material or extract is exposed to heat, light, or certain solvents.
Here's what makes this important: Cannabis plants don't naturally produce THC or CBD in their active forms. Instead, they biosynthesize acidic precursors – THCA, CBDA, and CBGA. These acidic cannabinoids must be decarboxylated to produce the effects consumers expect.
The basic reaction:
THCA → THC + CO₂
This reaction is thermally driven and irreversible under normal conditions, making precise temperature control essential for achieving your desired cannabinoid profiles.

The Chemistry: Mass Balance and Conversion Factors
When the carboxyl group is removed, molecular weight decreases. This mass loss must be accounted for in yield calculations and quality control testing.
The decarboxylation factor is 0.877 × THCA value by weight.
Here's a simple quality control check: Weigh your oil before decarboxylation, then weigh it again afterward. Your starting weight × 0.877 should be close to your ending weight. If there's a significant discrepancy, you're losing cannabinoids to thermal degradation.
Table 1: Cannabinoid Molecular Weight Changes During Decarboxylation
| Cannabinoid Pair | Acidic MW (g/mol) | Neutral MW (g/mol) | Mass Loss (%) |
|---|---|---|---|
| THCA → THC | 358.5 | 314.5 | 12.3% |
| CBDA → CBD | 358.4 | 314.5 | 12.3% |
| CBGA → CBG | 360.5 | 316.5 | 12.2% |
Note: The 0.877 factor (100% – 12.3% = 87.7%) reflects the theoretical maximum conversion. Actual yields depend on process conditions and material quality.
Decarboxylation Kinetics: Why Temperature Matters
Research by Wang et al. (2016) established that cannabinoid decarboxylation follows first-order reaction kinetics. The rate of conversion depends on temperature according to the Arrhenius equation – higher temperatures yield faster decarboxylation.
Critical insight: THCA decarboxylates approximately 2-3 times faster than CBDA at equivalent temperatures. This is why CBD-dominant materials require longer processing times than THC-dominant materials.
Table 2: Activation Energies by Cannabinoid
| Cannabinoid | Relative Rate | Activation Energy | Process Implication |
|---|---|---|---|
| THCA-A | 1.0 (reference) | 85-90 kJ/mol | Fastest conversion |
| CBDA | 0.3-0.5× | 110-115 kJ/mol | Requires a longer time |
| CBGA | 0.35-0.5× | 105-110 kJ/mol | Similar to CBDA |
When to Decarboxylate: Before or After Extraction?
This is one of the most important decisions in cannabis processing, and it significantly impacts your workflow and product flexibility.
Pre-extraction decarboxylation:
- Changes the polarity of cannabinoid molecules (acid form converts to neutral form)
- Lowers polarity and dielectric constant
- Enables non-polar solvents to extract more efficiently
- Drawback: You cannot produce acidic cannabinoid products from that material
Post-extraction decarboxylation:
- Preserves flexibility – the same crude extract can be processed into THC/CBD products OR preserved as THCA/CBDA products
- Allows you to respond to market demand
- Enables quality testing before and after conversion
Practical Decarboxylation Temperatures and Times
The optimal temperature and time depend on your starting material (plant vs. extract) and target cannabinoid (THC vs. CBD).
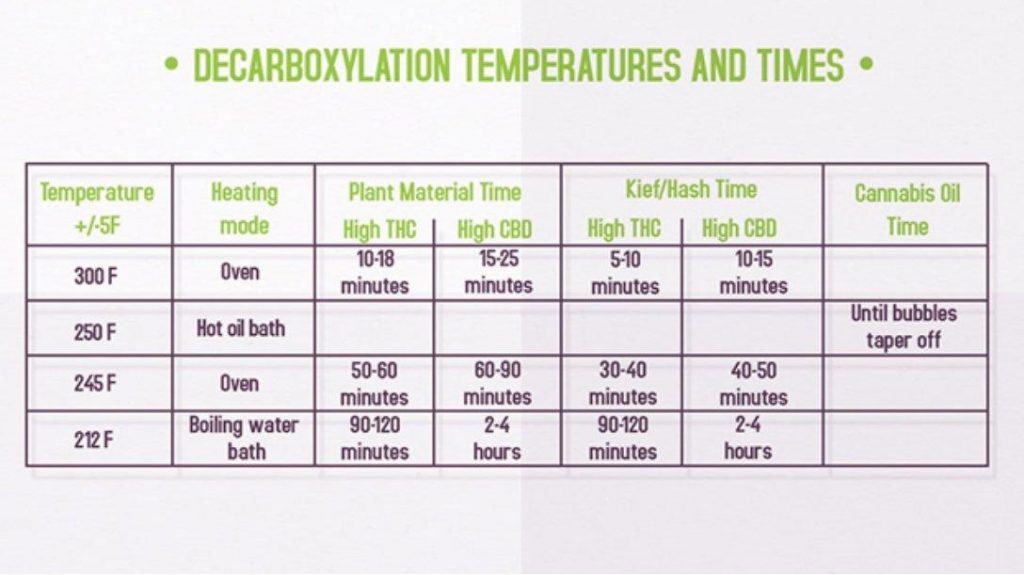
Figure 2: Decarboxylation Temperatures and Times for Various Heating Methods
Key Observations:
- High-THC materials decarboxylate faster than high-CBD materials at all temperatures
- Kief and hash require shorter times than plant material due to higher cannabinoid concentration
- Cannabis oil decarboxylation is monitored by bubbling – the process is complete when bubbles taper off
- Lower temperatures require longer times but preserve more terpenes
Oil Decarboxylation Challenges
Decarboxylating extracted oil is more difficult to control than flower decarboxylation. The rate depends on multiple factors: volume, temperature, and heat-exchange efficiency within the vessel.
Due to these variables, loss rates are typically higher. Standard oil decarboxylation typically achieves only 88% efficiency, meaning 12% of cannabinoids are lost beyond the theoretical CO₂ release.
PURE5™ has developed a low-temperature decarboxylation solution that achieves 90%+ efficiency, significantly reducing product loss compared to conventional methods.
Why Vacuum Ovens Are Superior for Decarboxylation
Vacuum ovens provide a controlled environment that prevents oxidation and enables lower operating temperatures:
- Oxygen elimination prevents oxidative degradation
- Lower effective temperatures reduce thermal stress on cannabinoids and terpenes
- Uniform heating eliminates hot spots that cause localized degradation
- CO₂ removal shifts equilibrium toward products, improving conversion efficiency
Pro tip: Monitor the vacuum gauge during decarboxylation. When it settles to zero, CO₂ release is complete and your material is fully decarboxylated.
How Extraction Method Affects Decarboxylation
Your extraction method fundamentally determines the cannabinoid profile of your crude extract. High-temperature methods cause partial, uncontrolled decarboxylation, while room-temperature methods preserve acidic forms entirely.
Table 3: Extraction Methods and Decarboxylation Impact
| Method | Temp Range | Decarb During Extraction | Post-Processing Required |
|---|---|---|---|
| Supercritical CO₂ | 88-140°F (31-60°C)+ | Partial (variable) | Winterization + controlled decarb |
| Ethanol | -40 to 172°F (-40 to 78°C) | Variable (temp dependent) | Evaporation + decarb |
| Hydrocarbon | -40 to 32°F (-40 to 0°C) | None | Purge + decarb |
| R134a (PURE5™) | 68-82°F (20-28°C) | None | Optional controlled decarb |
The Room-Temperature Advantage
Room-temperature extraction using R134a provides unique advantages:
- Product flexibility: Same crude can become THC/CBD products OR THCA/CBDA products
- Precise control: Decarboxylation parameters optimized for specific requirements
- Terpene preservation: 95-99% retention vs. 20-55% for high-temperature methods
- Quality verification: Full potency testing possible before and after decarboxylation
Post-Processing Decision Tree
Understanding what post-processing steps are required after extraction is critical for facility planning and cost analysis.
Table 4: Post-Processing Requirements by Extraction Method
| Extraction Method | Winterization | Solvent Purge | Decarb Required | Distillation | Total Steps |
|---|---|---|---|---|---|
| Supercritical CO₂ | YES — waxes extracted | Minimal | YES — partial conversion | Often needed | 3-4 steps |
| Ethanol (Warm) | YES — fats/waxes | YES — rotovap | YES | Often needed | 4 steps |
| Ethanol (Cryo) | Sometimes | YES — rotovap | YES | Often needed | 3-4 steps |
| Hydrocarbon (BHO) | NO — selective | YES — vacuum purge | YES | Optional | 2-3 steps |
| R134a (PURE5™) | NO — selective | NO — food-grade | OPTIONAL | Optional | 0-1 steps |
Key Insight: Each additional post-processing step increases equipment costs, labor time, potential yield loss, and quality degradation risk. R134a extraction minimizes or eliminates these steps, resulting in faster time-to-market, lower operational costs, and higher-quality final products.

Terpene Preservation During Decarboxylation
Terpenes are volatile compounds that contribute to cannabis aroma, flavor, and therapeutic effects through the entourage effect. Their low boiling points make them vulnerable to loss during decarboxylation.
Table 5: Common Cannabis Terpenes and Decarboxylation Risk
| Terpene | Boiling Point | Primary Aroma | Risk at Decarb Temps |
|---|---|---|---|
| α-Pinene | 311°F (155°C) | Pine | High (>248°F/120°C) |
| Myrcene | 333°F (167°C) | Earthy, musky | High (>266°F/130°C) |
| Limonene | 349°F (176°C) | Citrus | Moderate (>284°F/140°C) |
| Linalool | 388°F (198°C) | Floral | Moderate (>302°F/150°C) |
| β-Caryophyllene | 514°F (268°C) | Spicy, woody | Very low |
| Humulene | 388°F (198°C) | Hoppy | Moderate |

Terpene Preservation Strategies
- Lower temperature, longer time: Using 230-250°F (110-121°C) instead of 284°F (140°C) preserves more terpenes
- Vacuum processing: Reduces boiling points, allowing lower operating temperatures
- Terpene separation before decarb: Extract terpenes first, decarboxylate cannabinoids, then recombine
- Sealed systems: Prevent terpene vapor escape during heating
Table 6: Terpene Retention by Extraction Method
| Method | Terpene Retention | Primary Degradation Pathway |
|---|---|---|
| Supercritical CO₂ | 20-45% | Thermal + Oxidative |
| Ethanol (Warm) | 25-40% | Thermal + Solvent recovery |
| Ethanol (Cryo) | 40-55% | Thermal during recovery |
| Hydrocarbon (BHO) | 60-75% | Purge degradation |
| R134a (PURE5™) | 95-99% | Minimal/None |
Process Economics: The Real Cost of Decarboxylation
Understanding yield loss during decarboxylation is critical for accurate production planning and cost analysis.
Theoretical mass loss (unavoidable): 12.3% from CO₂ release (the 0.877 conversion factor)
Process losses (controllable): Additional losses from thermal degradation, oxidation, and volatilization. Standard oil decarboxylation achieves only 88% efficiency. PURE5™ low-temperature solutions achieve 90%+ efficiency.
Table 7: Total Cost of Ownership Comparison
| System | Extraction CapEx | Facility CapEx | Total CapEx | Terpene Retention |
|---|---|---|---|---|
| CO₂ + Decarb | $450,000 | $50,000 | $500,000 | 20-45% |
| Ethanol + Decarb | $125,000 | $100,000 | $225,000 | 25-40% |
| Hydrocarbon + Decarb | $200,000 | $150,000 | $350,000 | 60-75% |
| R134a + Optional Decarb | $125,000 | $0 | $125,000 | 95-99% |
Note: Facility CapEx includes C1D1 room construction for flammable solvents, high-pressure rated rooms for CO₂, and ventilation systems. R134a systems require no special facility modifications.
Key Takeaways
- THCA decarboxylates faster than CBDA – use shorter times for THC products, longer for CBD
- The 0.877 factor (12.3% mass loss) is theoretical – actual efficiency depends on process control
- Vacuum ovens provide superior results – monitor the vacuum gauge settling to zero as the completion indicator
- Room-temperature extraction provides maximum flexibility and control over final cannabinoid profiles
- Standard decarb achieves 88% efficiency – PURE5™ solutions achieve 90%+ with reduced losses
- Terpene preservation requires attention – lower temperatures and sealed systems minimize loss
Frequently Asked Questions (FAQ)
For THC-dominant cannabis, 240°F (115°C) for 60 minutes in a vacuum oven provides optimal results. For CBD-dominant hemp, use 275°F (135°C) for 60 minutes due to CBDA's higher activation energy. Lower temperatures preserve more terpenes but require longer processing times. PURE5™ vacuum ovens maintain precise temperature control throughout the process, ensuring consistent results batch after batch.
For oil decarboxylation, watch for bubbling to taper off – this indicates CO₂ release is complete. When using a vacuum oven, monitor the vacuum gauge; when it settles to zero, decarboxylation is finished. You can also verify by weighing before and after – the final weight should be approximately 87.7% of the starting weight.
It depends on your product goals. Post-extraction decarboxylation preserves flexibility, allowing you to produce both activated (THC/CBD) and acidic (THCA/CBDA) products from the same crude. This is one of the key advantages of room-temperature R134a extraction – your crude comes out with acidic cannabinoids fully preserved, giving you complete control over your final product lineup.
CBDA has a higher activation energy (110-115 kJ/mol) compared to THCA (85-90 kJ/mol). This means CBDA requires more energy to initiate the decarboxylation reaction, resulting in slower conversion rates at equivalent temperatures. For hemp processors working with CBD-dominant material, this makes precise temperature control even more critical.
Theoretical mass loss from CO₂ release is 12.3% (reflected in the 0.877 conversion factor). Additional process losses from thermal degradation and oxidation vary by method. Standard oil decarboxylation achieves approximately 88% efficiency. PURE5™'s low-temperature decarboxylation solutions achieve 90%+ efficiency, reducing controllable losses and putting more product in your jars.
It can. Many terpenes have boiling points below common decarboxylation temperatures. α-Pinene and Myrcene are particularly vulnerable above 248-266°F (120-130°C). To preserve terpenes, use lower temperatures with longer times, vacuum processing, or consider extracting with a room-temperature method like R134a (which preserves 95-99% of terpenes) before performing controlled decarboxylation.
Flower decarboxylation is more straightforward with consistent results. Oil decarboxylation is harder to control because heat transfer depends on volume, vessel geometry, and stirring. Oil decarb typically achieves only 88% efficiency compared to well-controlled flower decarb – which is why using professional-grade vacuum ovens with uniform heating is essential for commercial operations.
High-temperature extraction methods (like supercritical CO₂) cause partial, uncontrolled decarboxylation during the process – you lose control over your cannabinoid profile before post-processing even begins. Room-temperature methods like PURE5™ R134a extraction preserve acidic cannabinoids entirely, giving you full control over when and how decarboxylation occurs.
This factor accounts for the mass lost when CO₂ is released during decarboxylation. To calculate expected yield: multiply your starting THCA weight by 0.877 to get your theoretical THC yield. For example, 100g of THCA should produce approximately 87.7g of THC. Use this as a quality control check – if your actual yield is significantly lower, you're losing cannabinoids to thermal degradation.
The 12.3% CO₂ mass loss is unavoidable – that's the chemistry. But additional losses from thermal degradation can be minimized with the right equipment. Use vacuum ovens to eliminate oxygen, maintain consistent temperatures to avoid hot spots, and monitor the endpoint carefully to prevent over-processing. PURE5™ extraction combined with controlled vacuum oven decarboxylation delivers the highest total cannabinoid recovery in the industry.
Download the Complete Guide
This article covers the essentials of cannabis decarboxylation, but there's much more to learn. Our comprehensive eBook, “The Science of Cannabis Decarboxylation: A Technical Guide for Extraction Professionals“ includes:
- Detailed kinetics equations and scientific references
- Complete temperature/time charts for all material types
- In-depth extraction method comparisons
- Full process economics analysis
- Equipment recommendations and protocols
About PURE5™ Extraction
PURE5™ Extraction is a Phoenix, Arizona-based manufacturer of advanced cannabis extraction equipment utilizing R134a aerosol technology. Our systems operate at room temperature (68-82°F / 20-28°C) and low pressure (8-10 bar), achieving:
- 97% cannabinoid extraction efficiency
- 95-99% terpene preservation
- Non-flammable, FDA GRAS solvent
- No winterization required
- Low-temperature decarboxylation solutions with 90%+ efficiency
- Systems from 20L to 2000L capacity
For more information, visit www.pure5extraction.com or contact our team for a personalized consultation.
Follow us for daily tips, industry news, and exclusive offers:
References:
- “Decarboxylation Study of Acidic Cannabinoids: A Novel Approach Using Ultra-High-Performance Supercritical Fluid Chromatography” Wang et al., Cannabis and Cannabinoid Research, 2016
- “Analysis of Cannabinoids in Commercial Hemp Seed Oil and Decarboxylation Kinetics Studies of Cannabidiolic Acid (CBDA)” Citti et al., Journal of Pharmaceutical and Biomedical Analysis, 2018
- “Solubility of Δ9-Tetrahydrocannabinol in Supercritical Carbon Dioxide: Experiments and Modeling” Perrotin-Brunel et al., The Journal of Supercritical Fluids, 2010





