Abstract
The medicinal potential of the natural chemicals contained in plants is unlocked via the intricate and interesting process of plant extraction. This comprehensive article delves into the various plant extraction methods and modern separation techniques utilized to isolate and purify valuable plant-derived substances. From traditional maceration and decoction to cutting-edge supercritical fluid extraction and simulated moving bed chromatography, each method plays a crucial role in revealing the bioactive secrets hidden within botanical sources.
Table of Contents
ToggleIntroduction:
The study of botanical extraction has a great deal of potential for finding new bioactive substances that will transform industry business and medicine. By extracting and purifying these natural substances, researchers can explore their potential therapeutic applications. This article explores the intricate process of plant extraction and illuminates the many methods used to produce pure and effective natural goods.
What is plant extract? Simply put, it is the concentrated substance obtained from plants through various extraction methods, each designed to isolate and retain the beneficial bioactive compounds. In this article, we detail the extraction of plant extracts procedure-explaining step by step how each method is used to release and purify the potent natural compounds of plants.
Extraction Methods for Natural Products:
Maceration:
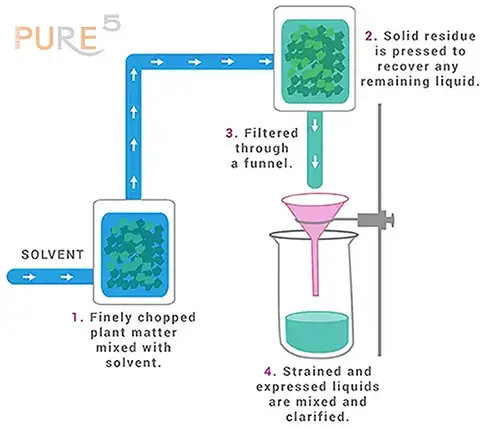
Maceration, also known as infused oils, is a process that extracts therapeutic properties from plant material using carrier oils as solvents. The plant material is finely cut and placed in a closed vessel along with a solvent. After allowing it to stand for a week, the mixture is occasionally shaken to ensure proper extraction. The liquid is then strained to separate the infused oil from the solid residue, and any remaining liquid is recovered by pressing the residue. By adding 5% Vitamin E oil or Wheatgerm oil, the infused oil's shelf life is enhanced, preventing rancidity. For a clear and pure oil, the liquids can be clarified through filtration or subsidence. This maceration process captures more of the plant's essence, making it an ideal choice for retaining valuable offerings in the final product.
 Percolation:
Percolation:
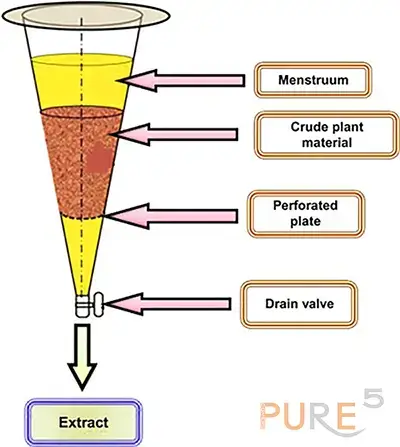
Percolation is a thorough extraction process that involves extracting the crude material with a new solvent in order to thoroughly remove all of the soluble components from a comminuted plant material. Repercolation involves reintroducing the percolate as the solvent, which lowers the amount of solvent used. Another development would be continuous countercurrent extraction, in which plant material and new solvent flow in the opposing directions to mix previously extracted material with new solvent. This quickens the extraction by increasing the gradient of extractives from the plant material to the solvent . In the fig below a schematic representation of the percolation-based extraction of a crude material is presented.
Decoction:
An infusion process that includes the boiling of plant material to extract water-soluble compounds. It is commonly used in traditional medicine systems worldwide.
Reflux Extraction:
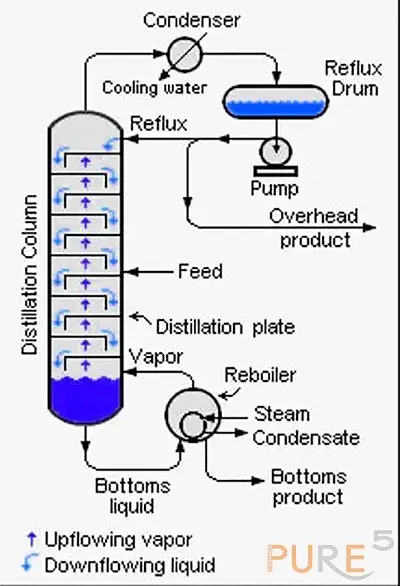
Reflux extraction ensures thorough extraction by continuously boiling and condensing the solvent, providing prolonged contact with the plant material. This method is appropriate for heat-stable compounds and enhances the efficiency of chemical extraction. A Reflux System principle is shown on the right.
Soxhlet Extraction:
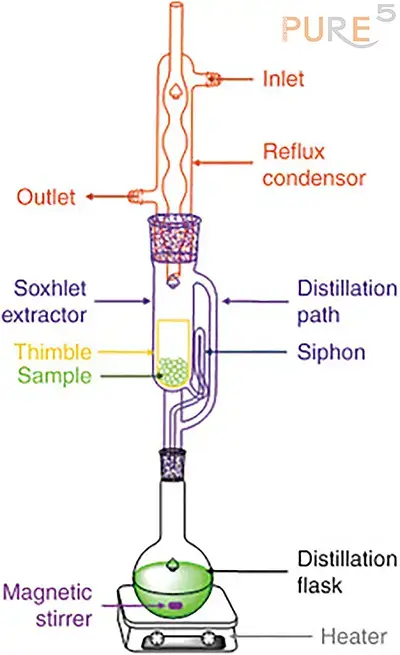
Pressurized Liquid Extraction (PLE):
PLE harnesses high pressure and temperature to accelerate extraction, making it highly versatile for extracting a wide range of compounds using various solvents. It is a preferred option in contemporary extraction techniques due to its effectiveness in extracting both polar and nonpolar molecules.
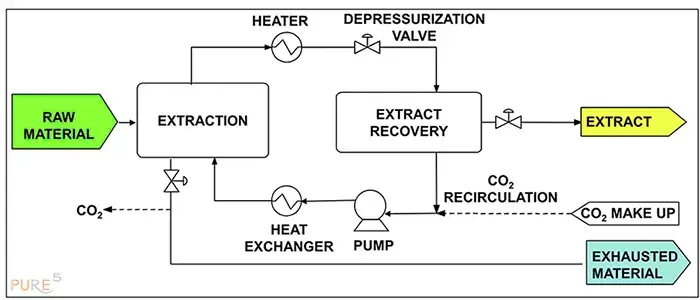
Supercritical Fluid Extraction (SFE):
There are 2 stages to any SFE procedure (see diagram in the next figure). The extractable components are first dissolved in the raw material as the supercritical CO2 passes through it. The extract-loaded solvent is then removed from the extractor and supplied into the separator. In the separator pressure is decreased and the solute is rendered insoluble, causing it to precipitate. As an alternative, the solute and supercritical solvent can be separated via a membrane, adsorption, or absorption. The solvent circuit can be used in this situation with practically continuous pressure.
Typically, the solid material needs to undergo pre-treatment. Water is frequently added to the process to make things simpler. This sum ranges from 10% to 40%. The source material might need to be ground to boost yield and extraction speed if the extracted solute is the desired product.
Ultrasound Assisted Extraction (UAE):
UAE harnesses ultrasonic waves to enhance extraction efficiency. The waves create tiny bubbles that rupture cell walls, facilitating the release of compounds. This non-thermal technique is quick and efficient at removing a variety of bioactive substances.
Microwave Assisted Extraction (MAE):
MAE speeds up the extraction process and is particularly effective for chemicals that are thermally labile by heating the solvent and plant material using microwave radiation. Its ability to significantly reduce extraction time makes it an energy-efficient technique.
Pulsed Electric Field Extraction (PEF):
PEF disrupts cell membranes using electric fields, releasing intracellular compounds without heat or solvents, preserving their natural state. This non-thermal technique works effectively for removing bioactive substances from delicate plant matter.
Enzyme-Assisted Extraction:
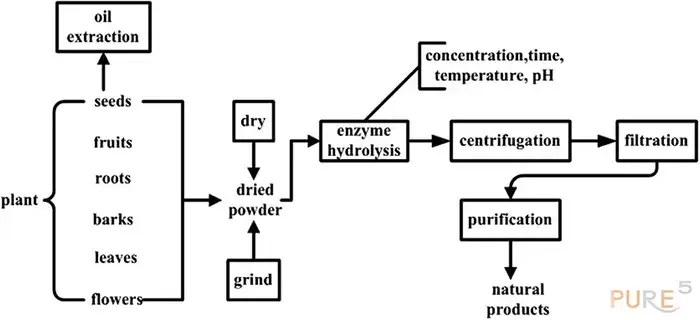
Enzyme-assisted extraction is a low-impact approach that weakens cell walls while increasing extraction selectivity. It is well-suited for complex plant matrices including difficult chemicals. Under moderate circumstances, enzymes are used to efficiently break down cell walls and extract bioactive substances. Extraction efficiency is determined by variables such as reaction temperature, extraction duration, pH, enzyme concentration, and substrate particle size. The diagram below depicts the Enzyme-Assisted Extraction technique for natural products.
Hydro Distillation and Steam Distillation:
These techniques capture volatile compounds by passing steam through plant material, yielding essential oils and aromatic compounds. Hydro distillation is suitable for extracting essential oils from plant material with high water content.
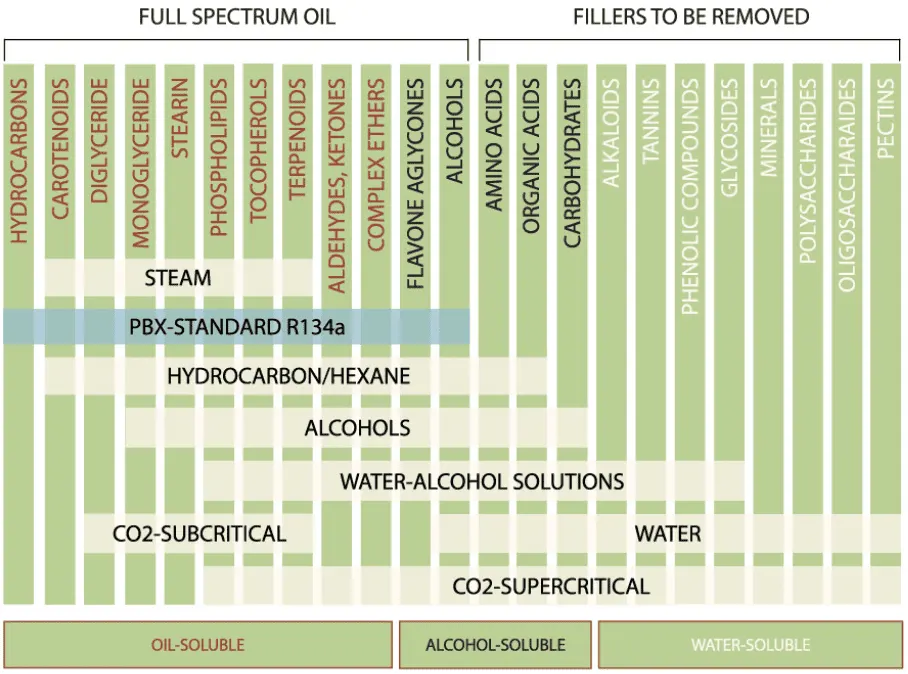
R134a Aerosol Extraction:
R134a is a safe and natural method that utilizes room temperatures and low pressure to extract plant compounds. It stands out for its ability to preserve thermally labile compounds and retain the valuable terpenes found in the plant being extracted. This environmentally friendly technique embraces nature's essence with elegance and ensures a pure and sustainable extraction process.
At PURE5™, we specialize in providing state of art plant extraction equipment, with a strong focus on R134a aerosol extraction. Our mission is to offer innovative and eco-conscious extraction solutions that harness the natural potency of botanical plants. With PURE5™, you can confidently embrace the natural path to extraction excellence. Select us for your extraction needs and experience the power of R134a Aerosol Extraction in the most efficient and sustainable way.
PURE5™ Extracted Oil:
How to create clean and potent plant oil in hours ready to use in products?
Separation Methods:
To isolate individual compounds from complex plant extracts, effective separation techniques are essential:
Separation Based on Adsorption Properties:
This method exploits varying affinities of compounds towards a stationary phase to separate them effectively. This category extensively employs chromatography techniques such as thin-layer and column chromatography.
Separation Based on the Partition Coefficient:
Compounds are separated based on their distribution between two immiscible phases, commonly achieved through liquid-liquid extraction. In this group, partition chromatography is one of the often employed methods.
Separation Based on Molecular Size:
Membrane Filtration (MF):
This technique separates compounds based on size and molecular weight using porous membranes, suitable for fractionation. It is ideal for separating compounds with a large molecular weight range.
Gel Filtration Chromatography (GFC):
GFC separates compounds based on size using a gel matrix, an ideal method for protein purification. It is valuable for isolating compounds with similar molecular weights.
Separation Based on Ionic Strength:
Utilizing differences in ionic strength, this approach separates compounds using techniques like Ion Exchange Chromatography. It is particularly useful for separating ionic compounds from a complex mixture.
Other Modern Separation Techniques:
Molecular Distillation (MD):
High-molecular-weight and thermosensitive chemicals can be separated by molecular distillation (MD) at low temperatures and vacuum, which helps to avoid deterioration. High-value chemicals and essential oils are frequently purified using this method.
Preparative Gas Chromatography (Prep-GC):
This method efficiently separates volatile compounds using modified gas chromatography equipment, perfect for preparative-scale separations. It may be used to separate rare and priceless substances from complicated combinations.
Supercritical Fluid Chromatography (SFC):
SFC method combines the benefits of liquid chromatography & gas chromatography, making it helpful for chiral separations & non-volatile chemical analysis. For the enantiomeric separation and purification of intricate combinations, it is a potent tool.
Molecular Imprinted Technology:
Molecularly imprinted polymers offer selective separation, providing high selectivity and cost-effective purification. It is a new method for separating target molecules from intricate matrices.
Simulated Moving Bed Chromatography:
A continuous separation method with reduced solvent consumption, ideal for large-scale processes. SMB chromatography is often employed in the food and pharmaceutical sectors to purify natural materials.
Multi-Dimensional Chromatographic Separation:
Enhancing separation efficiency through multiple columns with different stationary phases, this technique improves resolution and purification. For complicated mixes including a variety of chemicals, multi-dimensional chromatography is very useful.
Summary:
The process of extracting plants opens the door to discovering nature's hidden treasures. From traditional extraction methods rooted in ancient wisdom to cutting-edge technologies that leverage scientific advancements, each step plays a crucial role in isolating the therapeutic compounds found in plants. While plant extraction technology develops, it presents countless opportunities to harness the therapeutic power of these natural wonders for the advancement of the business industry and human health. Researchers can contribute to advancements in medicine and science by gaining a thorough understanding of extraction and separation techniques.
PURE5™ Extraction design and manufacture solventless extraction and distillation systems. The objective is to create a safe environment and simultaneously to elevate the customer experience. The systems provide high-quality output, speed, and economics for the entire process. The aerosol extracts are obtained at ambient temperature without degradation preserving the full oil vitality. The results are way better than any other extraction adopted for this market since none of those extracts the complete target active compounds.
Find more info on PURE5™'s equipment here.



 Percolation:
Percolation: